HANDY TIPS FOR H2 Chemistry – Chemical Energetics
Are you struggling with inorganic chemistry and finding it difficult to calculate accurately during the exams? This article is here to highlight important concepts in Chemical Energetics, which is mainly on energy changes during reactions, such as the different standard enthalpy changes, and exothermic and endothermic reactions. We hope this article can help you understand them better and provide you with some answering techniques to help you approach those questions better in your next examination.
Important concepts that are frequently tested:
- Endothermic and exothermic – Definitions and how the graphs look like!
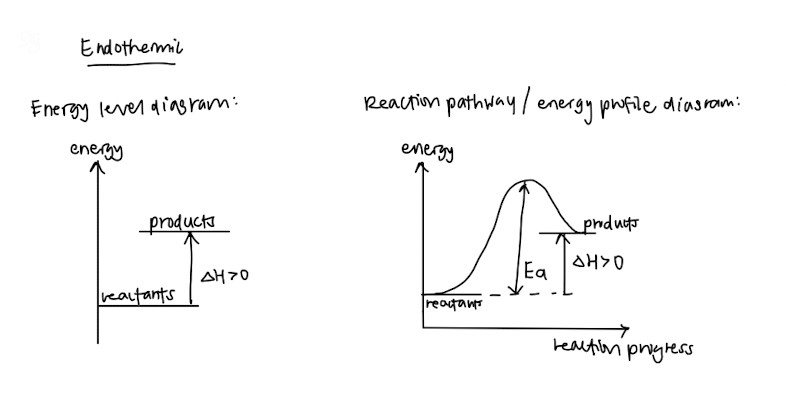
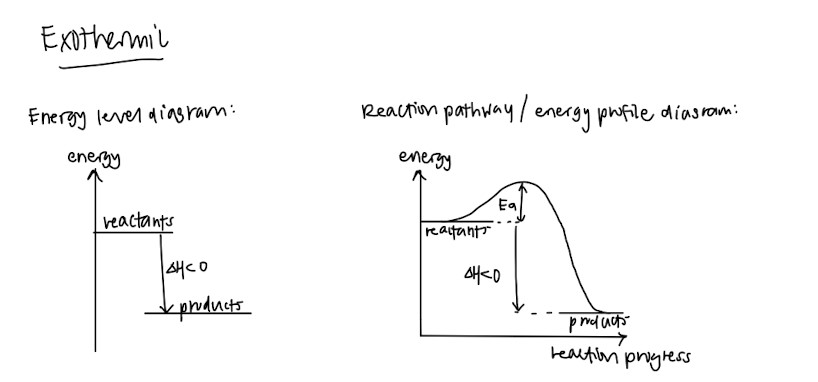
- Standard conditions and standard states (This should be standardised among schools but please double check) – They like to test this in MCQs or as a one mark question so please memorise these so you can get your free marks
– Temperature: 298K or 25 deg C
– Pressure: 1 bar = 10^5 Pa
– Concentration of any solution: 1.0 mol/dm3
– Standard states under standard conditions have zero enthalpy
Memorising the different enthalpy terms:
Of course, everything comes with practice, so don’t worry, the more you practise, the more you can commit these definitions to memory. To start off, remember most definitions end with the phrase “at 298K and 1 bar”. Substances are considered in the form of one mole, because you want to do calculations in its simplest form. The following list only includes the standard enthalpy changes that are frequently tested or confusing, but you should familiarise yourself with all the standard enthalpy changes in your syllabus.
Standard enthalpy change of formation
Simply put, the word formation makes remembering this definition very intuitive. Essentially, it is just the enthalpy change when 1 mole of substance is formed from its constituent elements in their standard states at 298K and 1 bar
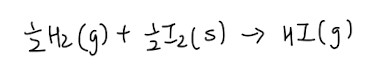
- By right, the enthalpy is supposed to be 0 kJ/mol
- If enthalpy is positive, compound is less stable
- If enthalpy is negative, compound is more stable
Standard enthalpy change of combustion
Burning always releases energy in the form of heat so the definition is heat evolved when 1 mole of substance is completely burnt in excess oxygen at 298K and 1 bar. You want combustion to be complete and in excess oxygen, otherwise other substances will be formed alongside.
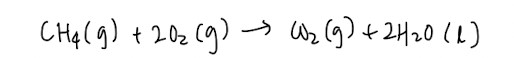
- Hess’ Law

- Energy level diagram
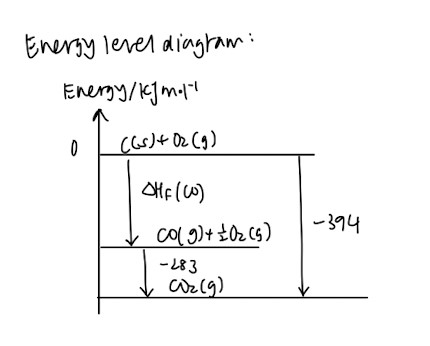
(Usually quite complicated at first, but it gets better when you familiarise yourself with the different definitions)
Bond dissociation energy
As the word “dissociation” suggests, you want to break something, and thus you need energy in order to do so, thus, bond dissociation will always be an endothermic process. As such, the definition is the energy required to break 1 mole of a particular covalent bond in a specific molecule in gaseous state.
- ΔH = sum of ΔH (bonds broken) – sum of ΔH (bonds formed)
- When calculating bond energies, remember to use the correct numbers. If you have multiple moles of a substance, you are supposed to multiply the bond energies by the number of moles of that substance (Students often lose marks here!)
Lattice Energy (LE)
It is the heat evolved when 1 mole of solid ionic compound is formed from its constituent gaseous ions. Since it is forming electrostatic forces of attraction between oppositely charged ions, LE always releases heat and is thus, always negative. Using this logic, the more negative LE is, the stronger the ionic bonding, and the more stable the ionic compound.
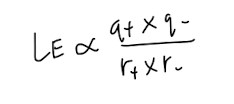
MCQs frequently test factors affecting lattice energy, so make sure you remember that and the equation for LE too! The factors affecting LE are:
- Ion charge
- Ion size/ size of inter-ionic distance
Ionisation Energy (IE)
First IE: energy required to remove 1 mole of electrons from 1 mole of gaseous atoms to form 1 mole of singly charged gaseous cations (The number of moles is very important, and remember to change the “singly charged” accordingly if the question is asking for second IE or third IE)
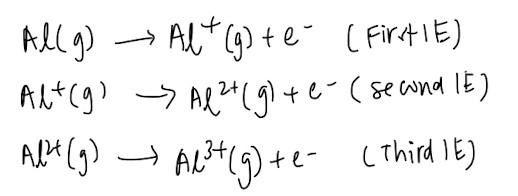
Electron Affinity (EA)
First EA: Enthalpy change when 1 mole of electrons is added to 1 mole of gaseous atoms to form 1 mole of singly charged gaseous anions
Relationship between LE, enthalpy changes of hydration and enthalpy changes of solution
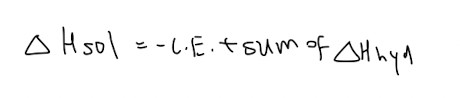
Gibbs Free Energy:
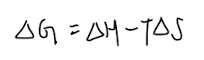
- When in doubt, use the equation to help you
- Remember the definition of entropy (measure of disorderliness/ randomness in the system)
- Factors that affect entropy
- Higher entropy: higher temperature, vaporisation/ melting, increase in gaseous particles, weaker intermolecular forces
- Lower entropy: dimerisation, water molecules hydrating ions are more orderly arranged than those in pure water
Very important concept that you should master: Born-Haber cycle (Energy level diagram especially)
- This is similar to the energy level diagram under the “Standard enthalpy change of combustion” section
- Make sure you look carefully at the number of moles involved in the reaction, because once you miscalculate the number of moles, you will calculate the LE wrongly and you will not get the full marks!
- Do more practices to familiarise yourself with the drawing of the energy level diagram and calculation
Questions with very standard answers:
- Why is there a higher experimental LE than theoretical: Higher experimental LE compensates for IE required
- What a large difference in experimental LE and theoretical LE mean: The ionic compound has covalent character.
- Why is 2nd IE higher than 1st IE: Further removal of electrons require more energy because shielding effect is reduced since there are less electrons.
- Why is 1st EA usually negative: The effective nuclear charge of the atom attracts the incoming electron. When there is a stronger attraction, the energy evolved is greater and LE is thus more negative.
- Why subsequent EA is always positive: Energy is required to overcome the electrostatic repulsion between the incoming electron and anion.
- Why a spontaneous reaction occurs at a slow rate or does not occur at all: Reaction requires high activation energy
- When the question asks why your calculated bond energy is different from the bond energy they give, remember the standard answer for this: Bond energy data in the data booklet are average values derived from a large range of molecules containing that particular covalent bond.
- Remember bond energy considers all molecules in gaseous state (common mistake among students)
Final words of advice
- Practice makes perfect! Keep practising energy-level diagram questions and calculations
- Don’t blindly memorise the standard enthalpy changes, try your best to understand the concept first before memorising them – it makes memorising a lot easier
If you need any help with H2 inorganic chemistry or other chemistry topics, our professional and very experienced tutors are always available to help.
Make an enquiry here and our friendly education specialists will be in touch with you shortly.
