
Handy tips for H2 organic chemistry
Are you struggling with organic chemistry and finding it difficult to answer organic chemistry questions in the exams? Organic chemistry is about the study of structures, properties of and reactions between carbon-containing compounds and is not as daunting as it seems! This article is here to highlight important concepts in organic chemistry, help you understand them better and provide you with some answering techniques to help you approach those questions better in your next examination. Note that this article does not include specific equations for chemical reactions because those are rather straightforward and can be mastered if you put in the effort to memorise and practise them.
Concepts used in questions that are easy to score:
- Hybridisations
- Questions like these usually test your knowledge of sp, sp2 and sp3 orbitals by asking you to draw out the hybrid orbitals for a given molecule, eg. CH3
- To answer these questions, you need to familiarise yourself with the type of bonds (sigma or pi, single or double), the bond angle and bond shape for the different types of hybridisations
- sp3: As the name suggests, there are 1 s orbital and 3 p orbitals. By adding them together, you get 4 single bonds, and knowing that single bonds are only sigma bonds, sp3 hybridisations consist of 4 single sigma bonds and are used for molecules that only contain single bonds, for example, CH4.
- sp2: Using the same logic as above, there are 3 single bonds in this hybridisation. However, you know that one carbon atom needs to be share all 4 of their valence electrons and 3 single bonds only require the sharing of 3 electrons. Hence, one of the bonds must be a double bond. Knowing that double bonds are pi bonds, sp2 hybridisations thus consist of 3 sigma bonds and 1 pi bond, 3 single bonds and 1 double bond. sp2 hybridisations are used for molecules that contain only one double bond, for example, C2H4
- sp: Using the same logic, there are 2 single bonds, but in order for carbon to share all 4 valence electrons, you need 2 double bonds. However, it is chemically unstable to have C=C=C to accomodate 2 double bonds, thus, you can only have a triple bond. sp hybridisation are only used for molecules with one triple bond, for exam C2H2
- Using hybridisation to explain basicity: for example, sp2 – more s character, lone pair of electron is closer to the nucleus and hence, less available for donation
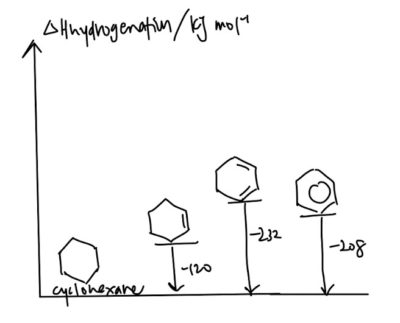
- Delocalisation energy: The more stable a compound is, the less negative ΔH(hydrogenation) is
- Formulae of organic compounds (very intuitive, just remember what each type of formula is) but remember to read the questions very carefully – some questions require you to use the displayed formula, skeletal formula or stereochemical formula, while some questions allow both.
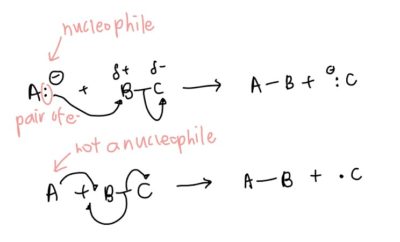
- Drawing movement of electrons: this is a super easy question to get marks easily from so don’t lose the opportunity!
- Full arrows (2 electrons): direction from anion (negative) to electropositive bonded atom – uses the logic unlike poles attract. Since the electropositive atom has been used to form bonds with the anion, the shared pair of electrons used to form the previous covalent automatically goes to the currently displaced atom (previously electronegative atom), forming an anion
- Half arrow (1 unpaired electron): In this case, electrons used for bonding with the unpaired electron come from the single bond.
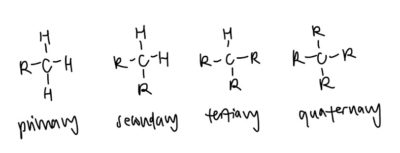
- Degree of substitution: another very straightforward concept
- Primary: One alkyl group bonded to main carbon atom
- Secondary: Two alkyl group bonded to main carbon atom
- Tertiary: Three alkyl group bonded to main carbon atom
- Quarternary: Four alkyl group bonded to main carbon atom
The next part will be covering the other 5 concepts that are easy to score, organic nomenclature, questions with very standard answers and some final words of advice.
If you need any help with topics such as H2 organic chemistry, Learners’ Lodge tutors are always available to help.
Make an enquiry here and our friendly education specialists will be in touch with you shortly.
