HANDY TIPS FOR H2 Chemistry – Equilibria
Are you struggling with inorganic chemistry and finding it difficult to calculate accurately during the exams? This article is here to highlight important concepts in Chemical Equilibria, which is the study of factors that disrupt the equilibrium, what happens when there is disequilibrium and how to bring the reaction back to dynamic equilibrium. This article will help you understand them better and provide you with some answering techniques to help you approach those questions better in your next examination.
Important concepts that are frequently tested:
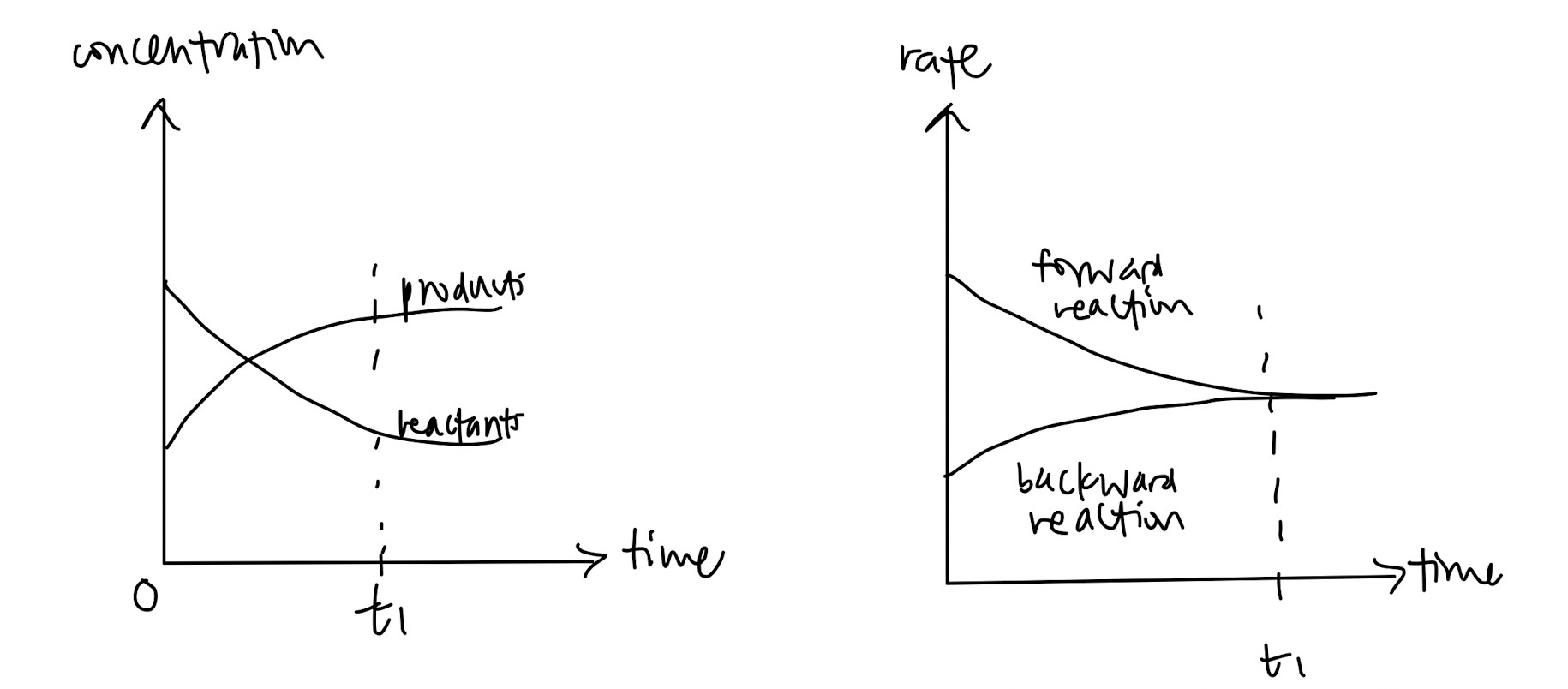
Definition of dynamic equilibrium – state of equilibrium where rate of forward reaction equals to rate of backward reaction (super easy to remember)
Knowing how to draw the concentration-time graph and rate-time graph
KC
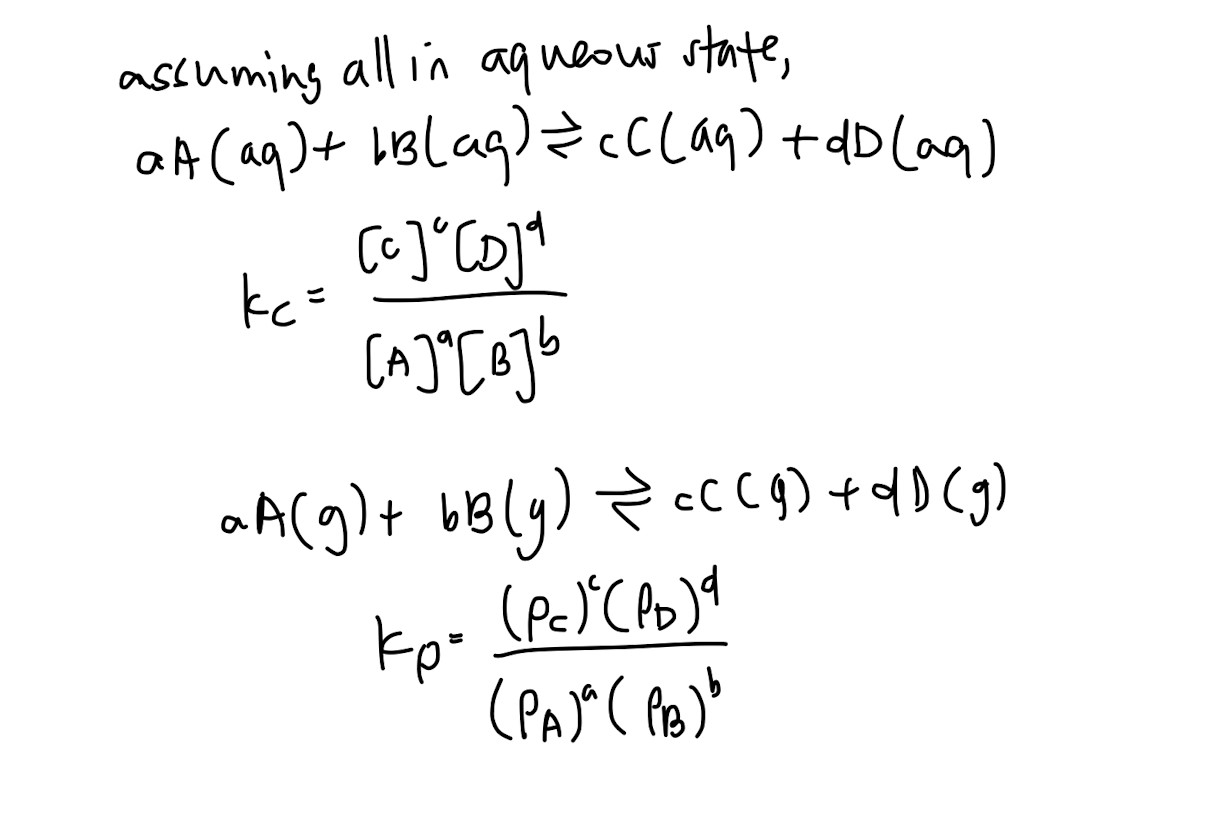
Kp (very similar to Kc)
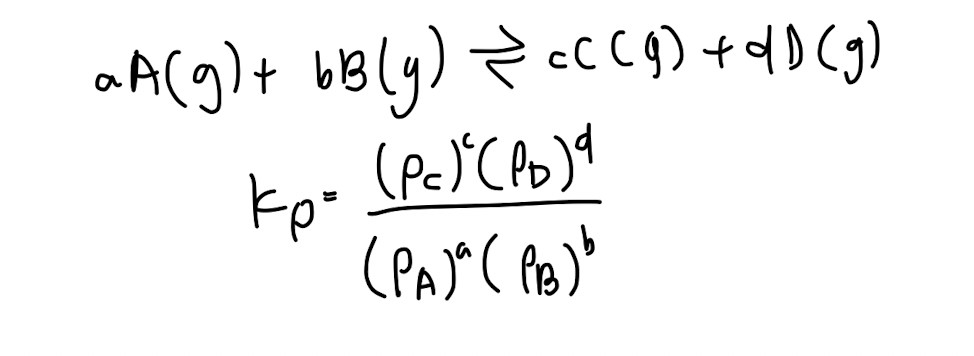
Kp and Kc are only affected by temperature (often tested to trick you)
Gibbs free energy: what does it mean when delta G < 0, delta G = 0, delta G > 0
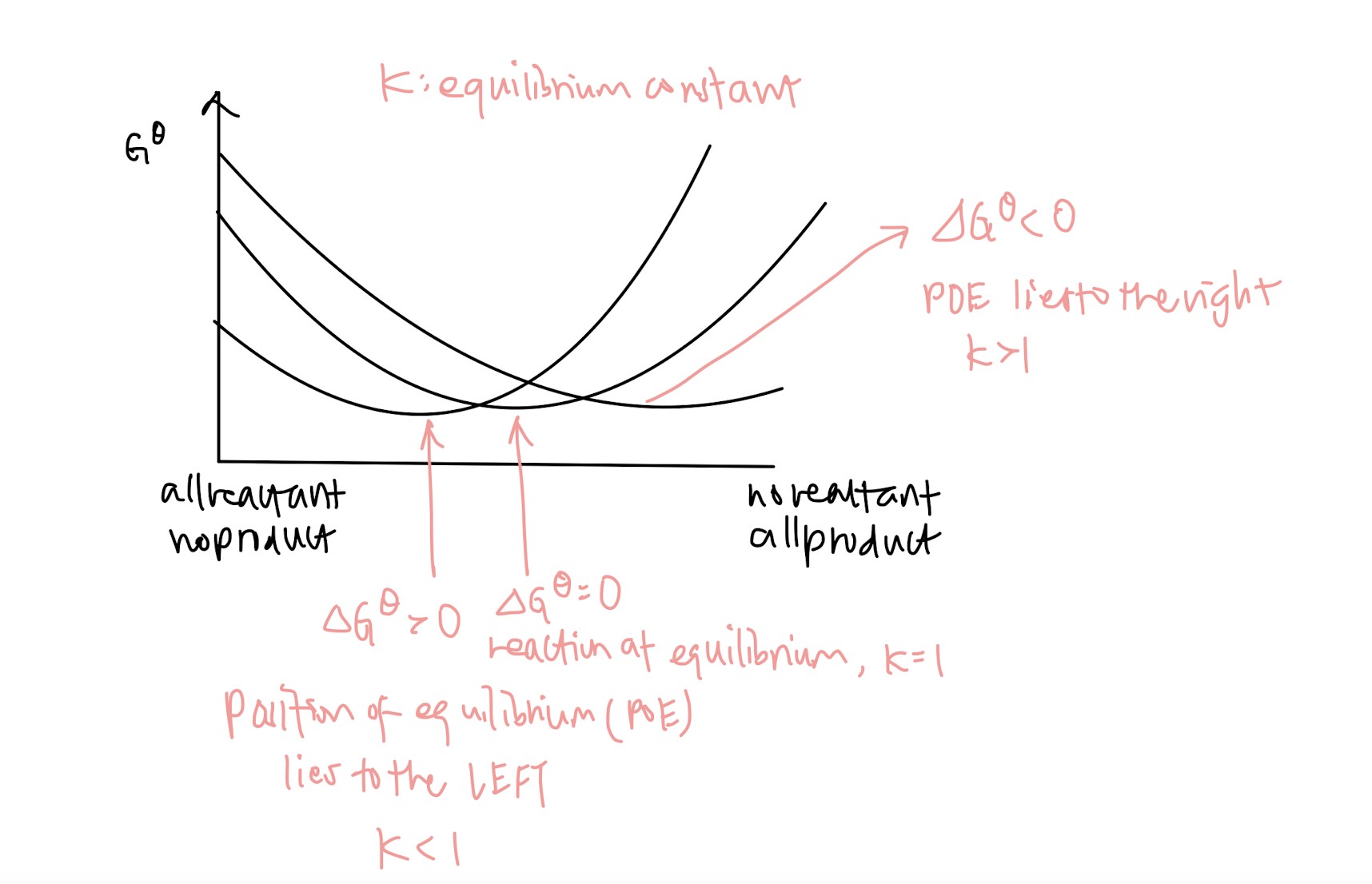
Definition of Le Chatelier’s Principle: If the conditions of a system in equilibrium are changed, the position of equilibrium moves to counteract that change (another easy mark)
How to draw the graph when concentration, temperature or pressure changes (Le Chatelier’s Principle is only affected by these three factors!!)
For example, if concentration increases,
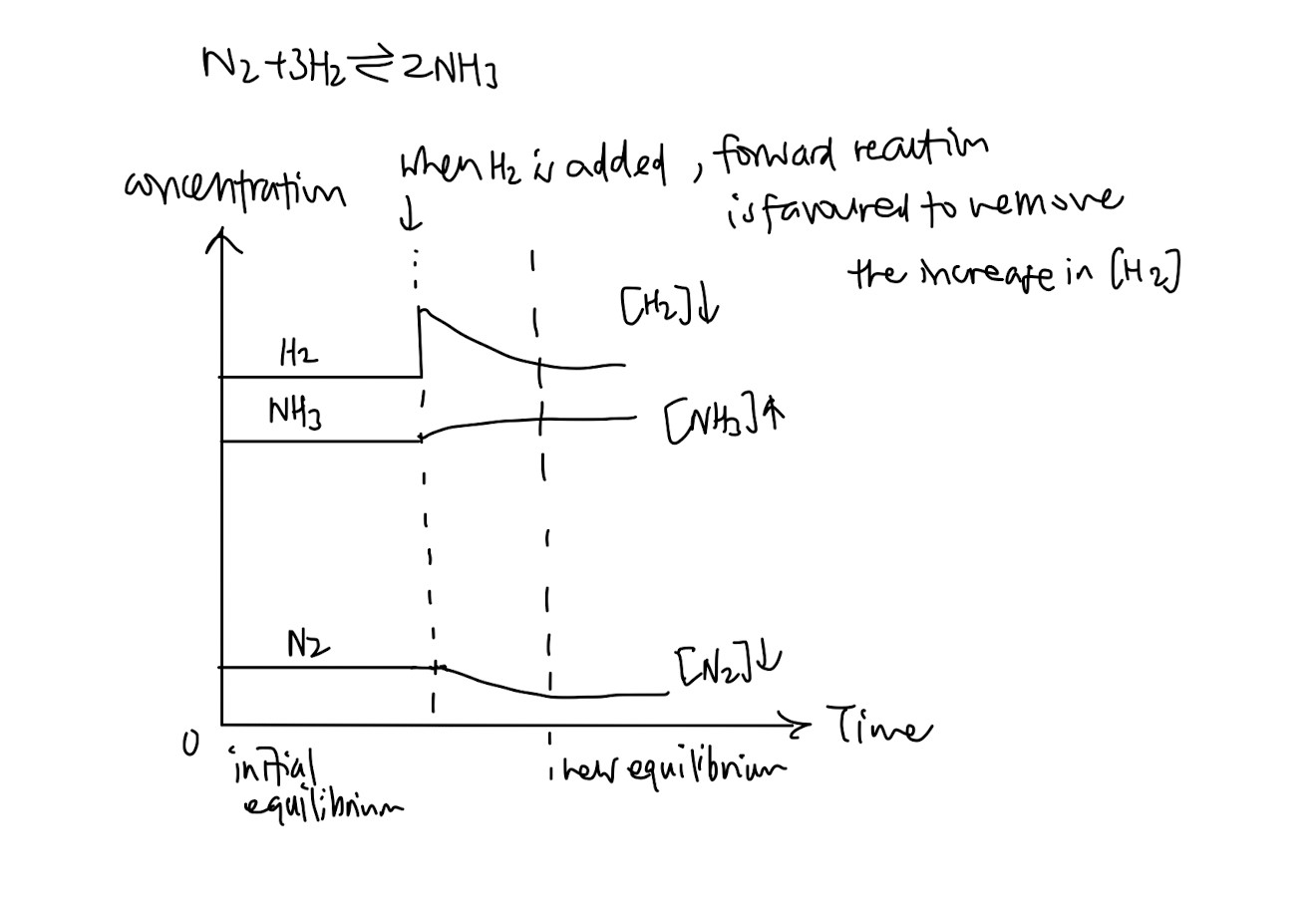
When in doubt, use the definition of Le Chatelier’s Principle to help you
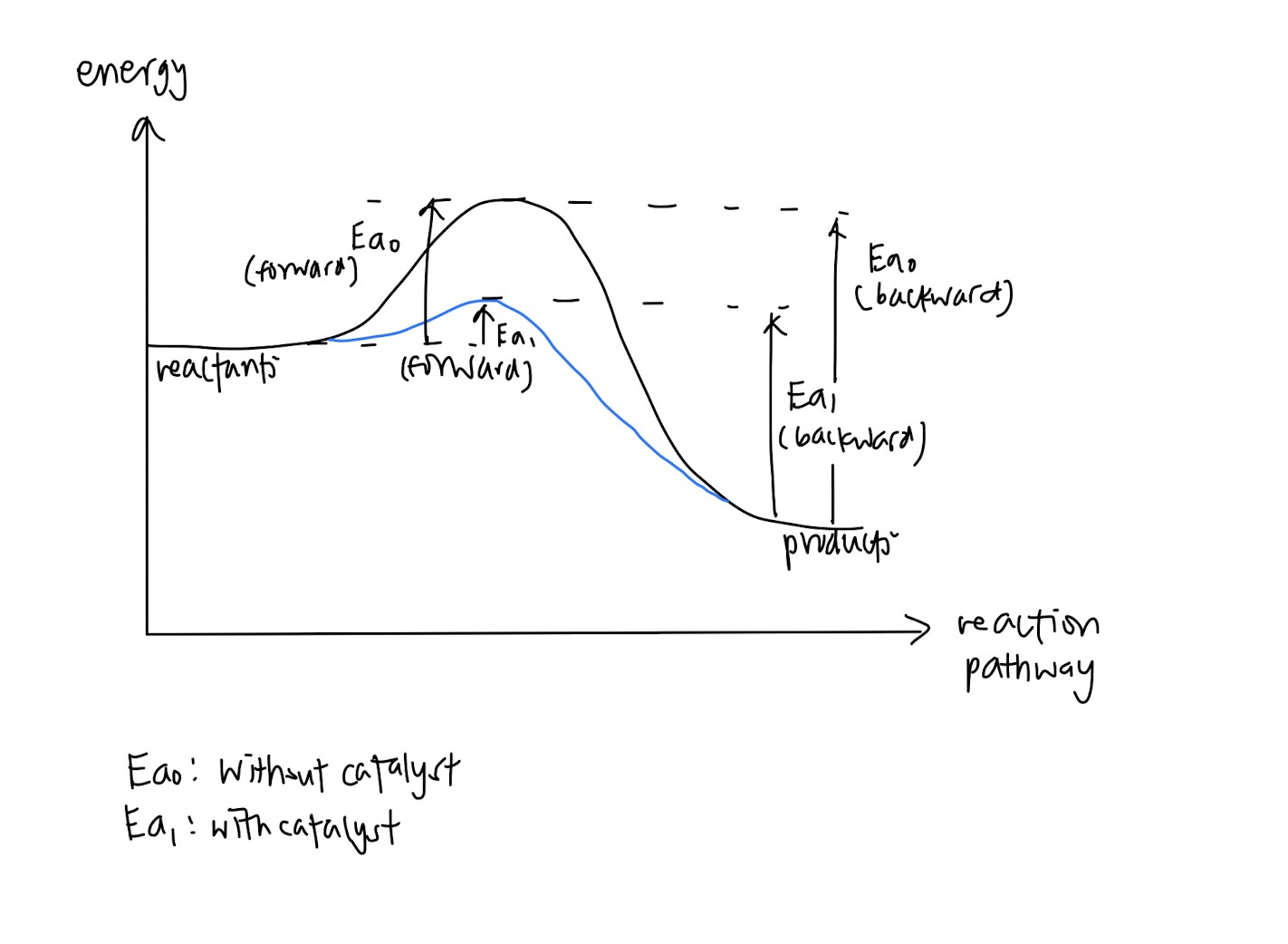
Effects of adding a catalyst
- Very simple, just remember catalyst is something that provides an alternative pathway with lower activation energy, and since less activation energy is needed, less time is needed for reaction to take place and rate of reaction decreases.
- This is not really tested but if they do, just remember that catalysts remain chemically unchanged
- Graph is important! Might be tested in MCQs or you may be asked to draw it out in paper 2 or 3
- Remember that catalyst never changes the position of equilibrium because they increase both the rate of forward reaction and backward reaction!! (often tested in MCQs) and since the catalyst does not change temperature, Kc does not change
Haber Process
- Remember the usual conditions for pressure and temperature (follow what your school suggests because different schools teach this differently)
- Catalyst is usually finely divided iron (because if iron pieces are too big, it will be less effective)
- Link it to the LCP:
- Pressure is high to favour forward reaction and increase yield but cannot be too high because that will require very strong pipes and vessels which will be more expensive
- Temperature is lower to favour forward reaction but cannot be too low either, or else the reaction will take very long and the process will not be cost-effective
Answering tips:
- When in doubt, always use the ICE table and you will be less likely to go wrong

- Remember to write the correct states of molecules in the equation!
Some concepts that may be harder to understand:
- When noble gas is added at constant pressure
- Use pV=nRT and partial pressure =
- When you add more molecules, in this case, noble gas molecules, total volume increases, but since the total pressure of the system stays constant, the proportion of total pressure with regards to each type of molecule decreases
- When noble gas is added at constant volume
- Use pV=nRT again
- When you add more molecules at constant volume, the total pressure increases (take the analogy of pumping more air into an enclosed container)
- However, since volume does not change, ratio of moles of each gas to the volume of the container will not change, hence partial pressure stays constant.
If you need any help with H2 inorganic chemistry or other chemistry topics, our professional and very experienced tutors are always available to help.
Make an enquiry here and our friendly education specialists will be in touch with you shortly.