Handy tips for H2 organic chemistry – Part 2
In part 1 of Learning H2 organic chemistry, you learn about concepts that are used in questions that are easy to score, including Hybridisations, Delocalisation energy, movement of electrons, and Degree of substitution. In this article, you will learn more about the other H2 organic chemistry concepts that usually come out during exam and are easy to score.
Some of the concepts include:
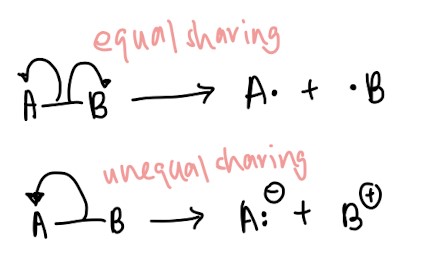
- Homolytic fission vs heterolytic fission: Use english to help you! Homo means same so homolytic fission means the pair of electrons is split evenly between two atoms. Use the same logic for heterolytic fission, hetero meaning different. ↓(Remember how to draw it!)↓
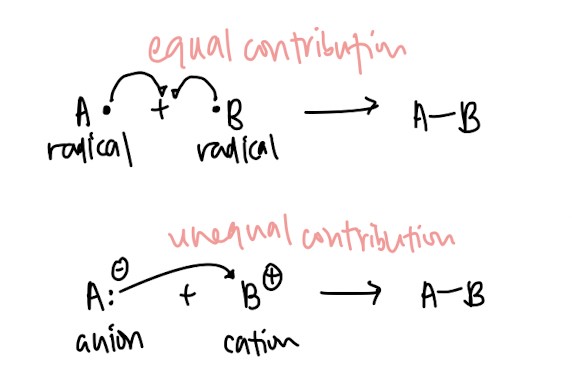
- Homolytic bond formation vs heterolytic bond formation: Same logic as above ↓(Remember how to illustrate it!)↓
- Free radical substitution: Memorise the mechanism and ↓how to draw it↓! It is commonly tested.
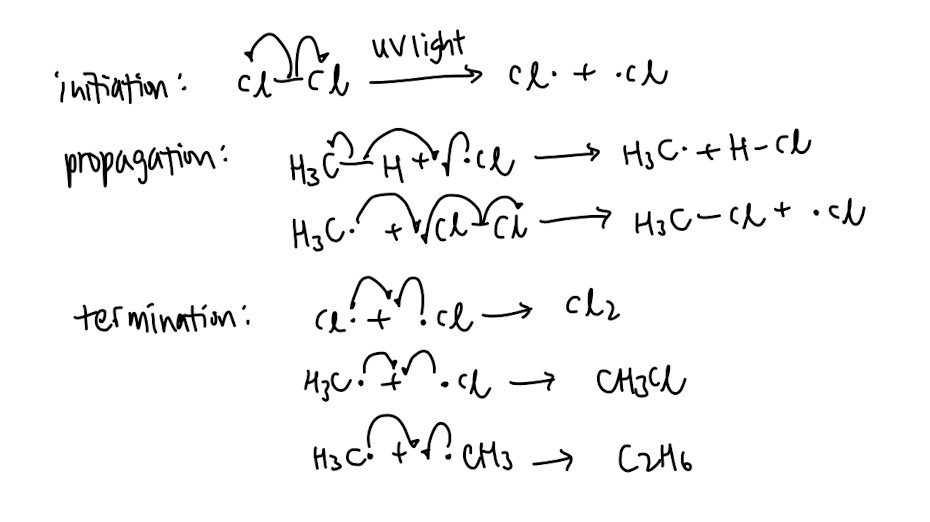
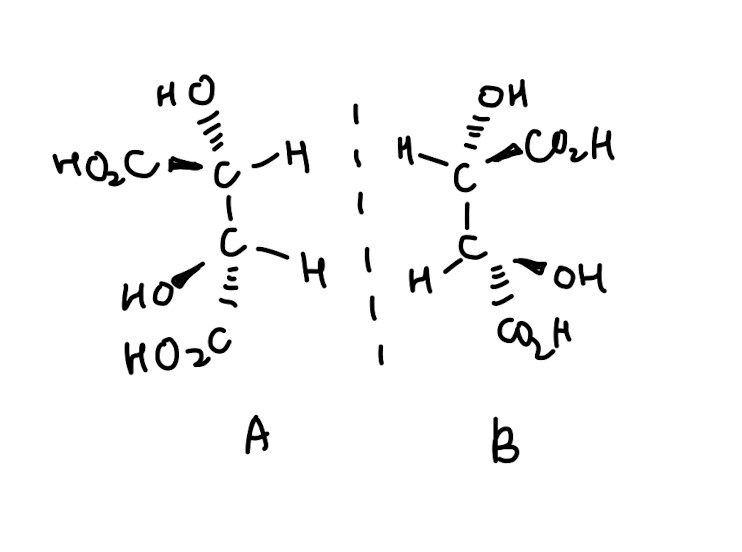
- Identifying and drawing stereoisomers, which are isomers that differ in the arrangement of atoms in space
- Aromatic compounds:
- Why aromatic compounds are only reactive towards electrophiles: The benzene ring is very electron-rich, thus it attracts electron deficient electrophiles/ repel electron-rich nucleophiles.
- Why aromatic compounds tend to undergo substitution: electrophilic addition disrupts aromaticity and destroys delocalisation; substitution keeps benzene ring unsaturated.
- Why OH- is used to generate a stronger nucleophile phenoxide ion: Because negative charge is more easily delocalised into benzene ring
- Why is there no electrophilic addition of the benzene ring: You want to maintain the aromatic structure and retain resonance stabilisation
Organic Nomenclature: Also an important concept because they may test you on naming but its not as hard as it seems. You don’t have to remember the order of functional group priorities entirely but just be aware of the prominent ones, and it will make more sense as you do more practices.
- If you see a carboxylic acid functional group, it will obviously become the suffix
- Parent nomenclature is the one with the longest main carbon chain
- Esters: The first part of the name is bound to the oxygen
Questions with very standard answers:
- Inductive effect
- Resonance effect (electron-withdrawing, electron-donating)
- Steric effects: Bulky alkyl group blocks path of electrophile
- Stereoisomerism: C=C cannot undergo free rotation unless pi bond is broken. The restricted rotation causes (compound) to be non-superimposable
- Why are A and B able to rotate plane-polarised light: They are chiral and enantiomers, which each enantiomer being optically active and able to rotate plane polarised light in equal but opposite directions
Difference between electrophilic and nucleophilic:
“-phile” means being attracted to something so using that logic, electrophile attracts electrons while nucleophiles donate their pair of electrons to the electrophile.
Memorising types of reactions: Use logic and practise drawing the reactions to memorise them efficiently!
Final words of advice
- Do more practices! The more you do, the more you will be able to grasp those concepts and commit the keywords to memory. In fact, as you practise, you will realise that questions are usually repetitive and test on the same few concepts.
- When memorising the different equations, as well as how to draw them, pay close attention to patterns. Once you identify those patterns, it will be much easier to understand how each reaction work and remember them better!
If you need any help with H2 organic chemistry or other chemistry topics, our professional and very experienced tutors are always available to help.
Make an enquiry here and our friendly education specialists will be in touch with you shortly.
